SCCO stands for small cubi-cuboctahedron (also non-hyphenated: small cubicuboctahedron). The SCCO is one of the 43 polyhedra collectively known as nonconvex uniform polyhedra (n-cUP). The mathematician John Lawrence Hudson illustrated the geometry and stellation of the SCCO in a recent article [1], pointing to the component forms (Platonic polyhedra) that are reflected in its name: six facial planes containing a square lying in the planes of a cube, eight facial planes containing a triangle lying in the planes of an octahedron, and six facial planes containing an octagon lying again in the planes of a cube. The latter cube is smaller than the first and lies therein with its facial planes parallel to corresponding planes of the first cube. The adjective “small” in SCCO distinguishes this solid from the “great” cubi-cuboctahedron.
The SCCO symmetry group: octahedral (also reflected in the name). Since the SCCO has three face types (equilateral triangle, square, and regular octagon), it has three different stellation diagrams.
Keywords: three-dimensional geometry, polytopes, solids, facial planes, 2-D facets
References and illustrations
[1] John Lawrence Hudson: Further Stellations of the Uniform Polyhedra. The Mathematical Intelligencer Fall 2009, Volume 31, No. 4, pp. 18-26.
[2] http://mathworld.wolfram.com/SmallCubicuboctahedron.html
[3] http://www.mathconsult.ch/showroom/unipoly/13.html
Thursday, December 17, 2009
Tuesday, December 15, 2009
The name dinosaur from the Greek words deinos (terrible) and sauros (lizard)
The name dinosaur (“terrible lizard”) was coined in the years after 1841 by the English biologist and palaeontologist Sir Richard Owen (1804-1892) [1,2]. Karl-Heinz Ludwig includes a small paragraph entitled Wie die Dinos zu ihrem Namen kamen (How the dinos got their name) in his book on climate change through the history of the Earth. Based on this German-language text [2] and information in Christopher McGowan's captivating book about prehistoric reptiles [1], the following summary can be shaped:
The word dinosaur, composed of the Greek words deinos (terrible) and sauros (lizard), first occurred in a publication by Richard Owen in 1842: Report on British fossil reptiles. Part II. Report of the British Association for the Advancement of Science, Plymouth, 11:60-204. By comparing fossil bones of Iguanodon (“iguana tooth”, an ornithopod), Megalosaurus (“big lizard”, a theropod) and Hylaeosaurus (“woodland lizard”, an ankylosaur), which had been discovered by William Buckland and Gideon Mantell in southern England, Owen recognized that these fossils represented a new group of Mesozoic reptiles. The classification of dinosaurs is not completed and the discovery of new dinosaur fossils and/or novel species can be expected to make news headlines any day.
Keywords: history, palaeontology, comparative anatomy, Mesozoic Era
References
[1] Christopher McGowan: Dinosaurs, Spitfires, & Sea Dragons. Harvard University Press, Cambridge, Massachusetts and London England, 1992 (first paperback edition); pages 190 and 343.
[2] Karl-Heinz Ludwig: Eine kurze Geschichte des Klimas • Von der Entstehung der Erde bis heute. Verlag C. H. Beck oHG, München, 2006; page 46.
The word dinosaur, composed of the Greek words deinos (terrible) and sauros (lizard), first occurred in a publication by Richard Owen in 1842: Report on British fossil reptiles. Part II. Report of the British Association for the Advancement of Science, Plymouth, 11:60-204. By comparing fossil bones of Iguanodon (“iguana tooth”, an ornithopod), Megalosaurus (“big lizard”, a theropod) and Hylaeosaurus (“woodland lizard”, an ankylosaur), which had been discovered by William Buckland and Gideon Mantell in southern England, Owen recognized that these fossils represented a new group of Mesozoic reptiles. The classification of dinosaurs is not completed and the discovery of new dinosaur fossils and/or novel species can be expected to make news headlines any day.
Keywords: history, palaeontology, comparative anatomy, Mesozoic Era
References
[1] Christopher McGowan: Dinosaurs, Spitfires, & Sea Dragons. Harvard University Press, Cambridge, Massachusetts and London England, 1992 (first paperback edition); pages 190 and 343.
[2] Karl-Heinz Ludwig: Eine kurze Geschichte des Klimas • Von der Entstehung der Erde bis heute. Verlag C. H. Beck oHG, München, 2006; page 46.
Thursday, November 26, 2009
Crime versus subculture: cracker versus hacker
A cracker is a person who gains oneself and/or other people illegal access to computers, most likely to perform some malicious activity. Often such a person is—incorrectly—called a hacker. A hacker's intention is not to damage a computer or to steal data for criminal undertakings. A hacker may detect security gaps and program errors hidden in operating systems, browsers and other software and bring those to public attention; but if so, then for their fixture. Typically, a hacker is a highly creative programmer with a lot of insider knowledge about computers and their inner workings. A hacker's programming style is explorative and unconventional, at least from a non-hacking viewpoint. Hackers are frequently seen (or see themselves) as part of a subculture. Some call it cyber punk.
Terms like password hacker or network hacker should be avoided, if they are meant to refer to malicious programming. Criminal activity is done by crackers—excluding nutcrackers!
Terms like password hacker or network hacker should be avoided, if they are meant to refer to malicious programming. Criminal activity is done by crackers—excluding nutcrackers!
Wednesday, November 25, 2009
Metonic cycle named after fifth-century B.C. astronomer Meton of Athens
The Greek astronomer Meton of Athens developed an effective system to adjust lunar calendars to the solar year [1]:
Keywords: astronomy, history, calendars, dating
References
[1] Jacqueline de Bourgoing: The Calendar • History, Lore, and Legend. Harry N. Abrams, Inc., Publishers, 2001; page 22.
[2] Eric W. Weisstein: Metonic Cycle.
[3] Tony Freeth: Decoding an Ancient Computer. Sci. Am. December 2009, 301 (6), pp. 76-83.
Meton supposedly observed that 19 solar years corresponded almost exactly to 235 lunar months. Since 19 lunar years consist of 228 months, it was thus necessary to add 7 intermediate months into 19 lunar years to put the lunar and solar calendars into phase.Before Meton, the Babylonians made similar (or the same) observations. Nevertheless, the 19-year, 235-lunar-month cycle is named Metonic cycle or Meton's cycle. It became the basis for the Greek calendar until the Julian calendar was introduced in 46 B.C. [2]. The Metonic cycle was adapted by the Jews in the 4th century A.D., who established a sophisticated lunisolar calendar. The Metonic cycle is still used today to determine the dates of the Jewish festival of Rosh Hashanah and the Christian festival of Easter [3].
Keywords: astronomy, history, calendars, dating
References
[1] Jacqueline de Bourgoing: The Calendar • History, Lore, and Legend. Harry N. Abrams, Inc., Publishers, 2001; page 22.
[2] Eric W. Weisstein: Metonic Cycle.
[3] Tony Freeth: Decoding an Ancient Computer. Sci. Am. December 2009, 301 (6), pp. 76-83.
Tuesday, November 24, 2009
Acronym in biochemistry: CALB for Candida antarctica lipase B
CALB stands for the enzyme lipase B specificly isolated from the yeast-like fungus Candida antarctica. CALB exhibits hydrolytic, ester synthesis and transesterification activity. It is a versatile biocatalyst, for example in asymmetric synthesis.
The structure and activity of CALB is currently investigated in various media including water, organic solvents, and ionic liquids as well as in solvent-free processing.
Keywords: organic chemistry, green chemistry, biocatalysis
Selected literature
[1] Kwang-Wook Kim, Boyoung Song, Min-Young Choi and Mahn-Joo Kim: Biocatalysis in Ionic Liquids: Markedly Enhanced Enantioselectivity of Lipase. Org. Lett. 2001, 3 (10), pp. 1507-1509. PDF-version.
[2] Francesco Secundo and Giacomo Carrea: Lipase activity and conformation in neat organic solvents. J. Mol. Catal. B: Enzym. December 2002, 19-20, pp. 93-102.
DOI: 10.1016/S1381-1177(02)00155-8.
[3] Anil Mahapatro, Ajay Kumar, Bhanu Kalra and Richard A. Gross: Solvent-free Adipic Acid/1,8-Octanediol Condensation Polymerizations Catalyzed by Candida antarctica Lipase B. Macromolecules 2004, 37, pp. 35-40. PDF-version.
[4] Hua Zhao, Cecil L. Jones and Janet V. Cowins: Lipase dissolution and stabilization in ether-functionalized ionic liquids. Green Chem. 2009, 11, pp. 1128-1138. DOI: 10.1039/b905388c.
The structure and activity of CALB is currently investigated in various media including water, organic solvents, and ionic liquids as well as in solvent-free processing.
Keywords: organic chemistry, green chemistry, biocatalysis
Selected literature
[1] Kwang-Wook Kim, Boyoung Song, Min-Young Choi and Mahn-Joo Kim: Biocatalysis in Ionic Liquids: Markedly Enhanced Enantioselectivity of Lipase. Org. Lett. 2001, 3 (10), pp. 1507-1509. PDF-version.
[2] Francesco Secundo and Giacomo Carrea: Lipase activity and conformation in neat organic solvents. J. Mol. Catal. B: Enzym. December 2002, 19-20, pp. 93-102.
DOI: 10.1016/S1381-1177(02)00155-8.
[3] Anil Mahapatro, Ajay Kumar, Bhanu Kalra and Richard A. Gross: Solvent-free Adipic Acid/1,8-Octanediol Condensation Polymerizations Catalyzed by Candida antarctica Lipase B. Macromolecules 2004, 37, pp. 35-40. PDF-version.
[4] Hua Zhao, Cecil L. Jones and Janet V. Cowins: Lipase dissolution and stabilization in ether-functionalized ionic liquids. Green Chem. 2009, 11, pp. 1128-1138. DOI: 10.1039/b905388c.
Sunday, November 22, 2009
Physical state of matter and class of particles named after Satyendranath Bose
Satyendranath Bose (also spelled Satyendra Nath Bose) was an Indian Physicist, born in 1894 in Calcutta (West Bengal, India) and later teaching physics at Dacca University in East Bengal (now Bangladesh). In 1924, Bose contacted Albert Einstein in Germany to help him publish a paper on quantum statistics that had been rejected by a British journal. Einstein translated the paper from English into German and submitted it to the journal Zeitschrift für Physik, in which the paper was then published with a note by Einstein. That was the beginning of the history of Bose-Einstein condensates. “Existing” as a predicted state of matter for more than fifty years, in 1989 Eric Cornell and his colleagues demonstrated its real existence by cooling rubidium atoms with laser light to nearly absolute zero temperature.
Bose's significant contributions to science are not only remembered with the terms Bose-Einstein statistics and Bose-Einstein condensate, but also with the name for a class of physical particles, the boson—a story beautifully told by John S. Rigden [2]:
References:
[1] Satyendranath Bose (1894-1974) at www-history.mcs.st-and.ac.uk/Mathematicians/Bose.html.
[2] John S. Rigden: Hydrogen • The Essential Element. Harvard University Press, Cambridge, Massachusetts and London, England, Third Printing 2003; page 234.
Bose's significant contributions to science are not only remembered with the terms Bose-Einstein statistics and Bose-Einstein condensate, but also with the name for a class of physical particles, the boson—a story beautifully told by John S. Rigden [2]:
There are various ways that fame comes to a scientist. For Satyendranath Bose it was asking Albert Einstein to run interference for him. Eventually his name was linked with Einstein's in both statistical method of dealing with quantum particles, called Bose-Einstein statistics, as well as the peculiar state of matter known as the Bose-Einstein condensate. In addition, Bose had a class of particles named after him: the boson. […]Keywords: physics, history, Bose-Einstein statistics, quantum chemistry
References:
[1] Satyendranath Bose (1894-1974) at www-history.mcs.st-and.ac.uk/Mathematicians/Bose.html.
[2] John S. Rigden: Hydrogen • The Essential Element. Harvard University Press, Cambridge, Massachusetts and London, England, Third Printing 2003; page 234.
Wednesday, November 18, 2009
Ludolph's number or Ludolphine number: pi
 For some time the transcendental number pi (π) was known in Germany as Ludolph's number or Ludolphine number. This name derived from the name of a German mathematician from Hildesheim, who emigrated to the Netherlands, where he taught fencing and mathematics in Delft and Leiden: Ludolph von Ceulen (1540-1610), also known by his latinized name Colonus. Ludolph van Ceulen (note the change from the German von to the Dutch van, meaning of or from in English) evaluated π to over 30 digits—long before the age of digital computing— by using the polygon method of Archimedes.
For some time the transcendental number pi (π) was known in Germany as Ludolph's number or Ludolphine number. This name derived from the name of a German mathematician from Hildesheim, who emigrated to the Netherlands, where he taught fencing and mathematics in Delft and Leiden: Ludolph von Ceulen (1540-1610), also known by his latinized name Colonus. Ludolph van Ceulen (note the change from the German von to the Dutch van, meaning of or from in English) evaluated π to over 30 digits—long before the age of digital computing— by using the polygon method of Archimedes.Keywords: mathematics, geometry, history, circle, radius, circumference, approximation of trancendental number
References:
R. M. Th. E. Oomes, J. J. T. M. Tersteeg and J. Top: Het grafschrift van Ludolph van Ceulen. Nieuw Arch. Wiskd. Juni 2000, 5/1 (nr. 2), pp. 57-62.
Internet: Ludolph's number and Ludolph van Ceulen.
Sunday, November 15, 2009
The unit of silence: a dirac
Several famous physicists have their name attached to a unit. Isaac Newton and his work on classical mechanics is memorized with the SI unit of force: the newton (symbol: N). From newton derives the unit of energy, Nm, which is named the joule (symbol: J) after James Prescott Joule. The list goes on. Paul Dirac, known for his work on the hydrogen atom and the spin of the electron and memorized with the Dirac delta function, also has “his” unit: a dirac (symbol unknown). But this is a unit of a somewhat different kind [1]:
Keywords: physics, units, history, humans, parents, childhood, solitude, silence, humor
Reference
John S. Rigden: Hydrogen • The Essential Element. Harvard University Press, Cambridge, Massachusetts and London, England, Third Printing 2003; p. 88-89.
Paul Dirac was a unusual person. Perhaps because Dirac's father demanded that his young son use French rather than his native English to converse with him, the young Dirac adopted the habit of silence during his childhood simply because he could not express his thoughts in French. Whatever the reason, the adult Paul Dirac was a man of silence. Dirac's silence was so intense that it inspired a little levity among physicists. In physics, the units given to physical quantities like time or length are important. Physicists, clearly in jest, have defined the unit of silence as the dirac.A few diracs—at least microdiracs— would suit everybody well at times!
Keywords: physics, units, history, humans, parents, childhood, solitude, silence, humor
Reference
John S. Rigden: Hydrogen • The Essential Element. Harvard University Press, Cambridge, Massachusetts and London, England, Third Printing 2003; p. 88-89.
Friday, November 13, 2009
Acronym in photovoltaics: LFC for laser fired contacts
Laser fired contacts (LFC) are electrically conductive point contacts on the rear of a solar cell, typically a cell based on crystalline silicon in which the silicon surface is separated from a metal layer by a dielectric passivation layer. LFC formation within such a structure is achieved by local laser heating, using a Nd-YAG laser which fires point contacts through the dielectric layer by metal/silicon alloying.
The LFC concept was developed by a team of researchers at the Fraunhofer Institute for Solar Energy Systems (ISE) in Freiburg, Germany [1].
Keywords: photovoltaics, solar cell design, device structure
Reference
E. Schneiderlöchner, R. Preu, R. Lüdermann and S. W. Glunz: Laser-fired contacts for crystalline solar cells. Progress in Photovoltaics: Research and Applications 2002, 10, pp. 29-34.
DOI: 10.1002/pip.422.
The LFC concept was developed by a team of researchers at the Fraunhofer Institute for Solar Energy Systems (ISE) in Freiburg, Germany [1].
Keywords: photovoltaics, solar cell design, device structure
Reference
E. Schneiderlöchner, R. Preu, R. Lüdermann and S. W. Glunz: Laser-fired contacts for crystalline solar cells. Progress in Photovoltaics: Research and Applications 2002, 10, pp. 29-34.
DOI: 10.1002/pip.422.
Thursday, November 12, 2009
Synonyms in polymer chemistry: constitutional repeating unit (CRU) and structural repeating unit (SRU)
A regular single-strand polymer consists of repeating units with two terminal groups at either end. The IUPAC Gold Book [1] defines a constitutional repeating unit (CRU) in a polymer as “the smallest unit the repetition of which constitutes a regular macromolecule, a regular oligomer molecule, a regular block or a regular chain.” The term structural repeating unit (SRU) is used synonymously for CRU [2]. A constitutional unit is an atom or group of atoms (with pendant atoms or groups, if any) comprising a part of the essential structure of a macromolecule, an oligomer molecule, a block or a chain [3].
References
[1] http://goldbook.iupac.org/C01286.html
[2] J. Kahovec, R. B. Fox and K. Hatada (Working Group): Nomenclature of Regular Single-Stranded Organic Polymers (IUPAC Recommendations 2002). Pure Appl. Chem. 2002, 74 (10), pp. 1921-1956. PDF-version.
[3] A. D. Jenkins, P. Kratochvíl, R. F. T. Stepto and U. W. Suter: Glossary of basic terms in polymer science (IUPAC Recommendations 1996). Pure Appl. Chem. 1996, 68 (12), pp. 2287-2311. PDF-version.
References
[1] http://goldbook.iupac.org/C01286.html
[2] J. Kahovec, R. B. Fox and K. Hatada (Working Group): Nomenclature of Regular Single-Stranded Organic Polymers (IUPAC Recommendations 2002). Pure Appl. Chem. 2002, 74 (10), pp. 1921-1956. PDF-version.
[3] A. D. Jenkins, P. Kratochvíl, R. F. T. Stepto and U. W. Suter: Glossary of basic terms in polymer science (IUPAC Recommendations 1996). Pure Appl. Chem. 1996, 68 (12), pp. 2287-2311. PDF-version.
Tuesday, November 10, 2009
English ‘naked’ and German ‘nackt’ in delicate circumstances
The English adjective naked is translated into German nackt. The words naked and nackt look and sound similar and linguistically have the same root. This suggests plain back-and-forth translation. However, there are notable exceptions as the following examples will illustrate.
The English phrase with the naked eye is typically not translated as mit dem nackten Auge. In German(y), one sees mit dem bloßen Auge (with the bare eye) or mit dem unbewaffneten Auge (with the unequipped eye).
The German term nackte Tatsache does not mean a naked fact, but may be translated as simple fact or brute fact.
In other contexts nackt may be best translated into English by taking adjectives such as nude, bare, plain, blunt or stark.
Keywords: English language, German language, translation, adjectives
The English phrase with the naked eye is typically not translated as mit dem nackten Auge. In German(y), one sees mit dem bloßen Auge (with the bare eye) or mit dem unbewaffneten Auge (with the unequipped eye).
The German term nackte Tatsache does not mean a naked fact, but may be translated as simple fact or brute fact.
In other contexts nackt may be best translated into English by taking adjectives such as nude, bare, plain, blunt or stark.
Keywords: English language, German language, translation, adjectives
Monday, November 9, 2009
LM for living morphine (alive and morphine given)
During the Vietnam War the letters L and M, written on the helmet of an injured soldier, indicated that he was still alive and had received an injection of morphine. In the Norwegian thriller Kakerlakkene (meaning cockroaches in English and Küchenschaben in German) by Jo Nesbø, the Vietnam veteran Ivar Løken has an LM history of its own. This thriller does not take place in Vietnam, but—years after the end of the Vietnam War—in Bangkok,Thailand. A war of a different kind is happening here. This war doesn't include an army, just a few people (and not all of them are Norwegians) connected via sex, gambling, and corruption.
Reference
Jo Nesbø: Kakerlakkene, originally published in Norwegian (Aschehoug, Oslo).
German-language edition: Kakerlaken. Ullstein Taschenbuch, Berlin, Juni 2009.
Reference
Jo Nesbø: Kakerlakkene, originally published in Norwegian (Aschehoug, Oslo).
German-language edition: Kakerlaken. Ullstein Taschenbuch, Berlin, Juni 2009.
Friday, October 30, 2009
Alcedo atthis, also known as Eurasian, European, or river kingfisher
Alcedo atthis is the systematic name of a bird otherwise known as common kingfisher or river kingfisher. The names Eurasian kingfisher or European kingfisher hint to the geographic range where Alcedo atthis is living, diving and fishing: in most of Europe and non-Siberian parts of Asia; but also in northern Africa and parts of Australia.
Its German name is Eisvogel, its French name is Martin-pêcheur, and its Spanish name is martín pescador . The adjectives gemein, commun, and común may respectively be added for common.
The kingfisher belongs to the Alcedinidae family (river kingfishers) in the suborder Alcedines of the order Coraciiformes (Ordnung Rackenvögel in German). A northern American relative is, for example, the belted kingfisher (Ceryle alcyon), whose habitats include rivers, lakes, coasts and the Sierra of California.
Keywords: ornithology, Coraciiformes
References
[1] Hannah Holmes: Blaze of Blue • The Eurasian kingfisher is flashy and feisty. National Geographic November 2009, Volume 216, Number 5, pp. 76-85.
[2] Grzimeks Tierleben • Neunter Band • Vögel 3 • Kindler Verlag AG, Zürich, 1970.
Its German name is Eisvogel, its French name is Martin-pêcheur, and its Spanish name is martín pescador . The adjectives gemein, commun, and común may respectively be added for common.
The kingfisher belongs to the Alcedinidae family (river kingfishers) in the suborder Alcedines of the order Coraciiformes (Ordnung Rackenvögel in German). A northern American relative is, for example, the belted kingfisher (Ceryle alcyon), whose habitats include rivers, lakes, coasts and the Sierra of California.
Keywords: ornithology, Coraciiformes
References
[1] Hannah Holmes: Blaze of Blue • The Eurasian kingfisher is flashy and feisty. National Geographic November 2009, Volume 216, Number 5, pp. 76-85.
[2] Grzimeks Tierleben • Neunter Band • Vögel 3 • Kindler Verlag AG, Zürich, 1970.
Monday, October 26, 2009
Acronym in chemistry: GDGT for glycerol dibiphytanyl glycerol tetraether
Glycerol dibiphytanyl glycerol tetraethers (GDGTs) are large cyclic tetraether molecules biosynthesized by archaebacteria as membrane building blocks, spanning the membrane lipid layer. Examples of GDGTs are caldarchaeol and crenarchaeol. The latter contains four cyclopentane and one cyclohexane ring within the “large-ring backbone” (see reference [1] and Crenarchaeol, derived from Crenarchaea). The use of biphytanyl glycerol diethers and GDGTs distinguish archaea from other bacteria and eukaryotes. Archaea live in extreme environments and the ether linkages, which are more stable than the ester linkages in membrane lipids of non-archaea, have been interpreted as an adaption to thrive or survive under extreme conditions.
The notation GDGT-n, in which n is an integer specifying the total number of cyclopentane rings in a GDGT molecules, has been introduced [1,2]. Caldarchaeol is a GDGT-0 and crenarchaeol a GDGT-4 compound.
Keywords: biochemistry, thermophilic organisms, pelagic crenarchaeota, membrane architecture
References
[1] J. S. Sinninghe Damsté, S. Schouten, E. C. Hopmans, A. C. T. Duin and J. A. J. Geenevasen: Crenarchaeol: the characteristic core glycerol dibiphytanyl glycerol tetraether membrane lipid of cosmopolitan pelagic crenarchaeots. J. Lipid Res. 2002, 43, pp. 1641-1651.
DOI: 10.1194/jlr.M200148-JLR200.
[2] J. S. Sinninghe Damsté, W. I. C. Rijpstra, E. C. Hopmans, F. G. Prahl, S. G. Wakeham and S. Schouten: Distribution of Membrane Lipids of Planktonic Crenarchaeota in the Arabian Sea. Appl. Env. Microbiol. 2002, 68, pp. 2997-3002.
DOI: 10.1128/AEM.68.6.2997-3002.2002.
The notation GDGT-n, in which n is an integer specifying the total number of cyclopentane rings in a GDGT molecules, has been introduced [1,2]. Caldarchaeol is a GDGT-0 and crenarchaeol a GDGT-4 compound.
Keywords: biochemistry, thermophilic organisms, pelagic crenarchaeota, membrane architecture
References
[1] J. S. Sinninghe Damsté, S. Schouten, E. C. Hopmans, A. C. T. Duin and J. A. J. Geenevasen: Crenarchaeol: the characteristic core glycerol dibiphytanyl glycerol tetraether membrane lipid of cosmopolitan pelagic crenarchaeots. J. Lipid Res. 2002, 43, pp. 1641-1651.
DOI: 10.1194/jlr.M200148-JLR200.
[2] J. S. Sinninghe Damsté, W. I. C. Rijpstra, E. C. Hopmans, F. G. Prahl, S. G. Wakeham and S. Schouten: Distribution of Membrane Lipids of Planktonic Crenarchaeota in the Arabian Sea. Appl. Env. Microbiol. 2002, 68, pp. 2997-3002.
DOI: 10.1128/AEM.68.6.2997-3002.2002.
Sunday, October 25, 2009
Crenarchaeol, derived from Crenarchaea
Crenarchaeol derives from Crenarchaea, linguistically and biochemically. Crenarchaea, a taxonomic grouping of archaea, are autotrophic organisms that are widespread in both surface and deep waters of the oceans [1].
Crenarchaeol, a glycerol dibiphytantyl glycerol tetraether (GDGT) that plays a critical role in the membrane structure of planktonic crenarchaea, was named in 2001 by Jaap Sinninghe Damsté of the Netherlands Institute of Sea Research on the North Sea Island of Texel [4-6], when he and colleagues isolated the compound and identified its structure using two-dimensional nuclear magnetic resonance spectroscopy [1]:
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; see, for example, pages 215-217 and glossary.
[2] Jack R. Holt: Introduction to the Kingdom Crenarchaea.
[3] Sue Barnsand Siegfried Burggraf: Crenarchaeota.
[4] J. S. Sinninghe Damsté, S. Schouten, E. C. Hopmans, A. C. T. Duin and J. A. J. Geenevasen: Crenarchaeol: the characteristic core glycerol dibiphytanyl glycerol tetraether membrane lipid of cosmopolitan pelagic crenarchaeots. J. Lipid Res. 2002, 43, pp. 1641-1651.
DOI: 10.1194/jlr.M200148-JLR200.
[5] J. S. Sinninghe Damsté, W. I. C. Rijpstra, E. C. Hopmans, F. G. Prahl, S. G. Wakeham and S. Schouten: Distribution of Membrane Lipids of Planktonic Crenarchaeota in the Arabian Sea. Appl. Env. Microbiol. 2002, 68, pp. 2997-3002.
DOI: 10.1128/AEM.68.6.2997-3002.2002.
[6] S. Schouten, E. C. Hopmans, E. Schefuß and J. S. Sinninghe Damsté: Distributional variations in marine crenarchaeotal membrane lipids: a new tool for reconstructing ancient sea water temperatures? Earth Planet Sci. Lett. 2002, 204, pp. 265-274. PDF-version.
Crenarchaeol, a glycerol dibiphytantyl glycerol tetraether (GDGT) that plays a critical role in the membrane structure of planktonic crenarchaea, was named in 2001 by Jaap Sinninghe Damsté of the Netherlands Institute of Sea Research on the North Sea Island of Texel [4-6], when he and colleagues isolated the compound and identified its structure using two-dimensional nuclear magnetic resonance spectroscopy [1]:
It [crenarchaeol] was distinguished from the other ring-containing tetraethers by a biphytanyl chain that had a six-carbon ring in addition to two of the usual five-carbon rings. Damsté christened the compound “crenarchaeol” and, noting that the six-carbon ring created a sort of bulge in the middle of the tetraether, teamed up with theoretical chemists from the University of Amsterdam to create a molecular model of an archaeal membrane structure with the new compound as core lipid. The result offered one possible solution to the apparent paradox of cold water crenarchaea: whereas the five-carbon rings made the biphytanyl chains fit together more snugly in a dense structure, addition of crenarchaeol with its bulging six-carbon ring kept them from packing together too closely and would, conceivably, allow the membrane to maintain its fluidity at low temperatures.References and interesting links
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; see, for example, pages 215-217 and glossary.
[2] Jack R. Holt: Introduction to the Kingdom Crenarchaea.
[3] Sue Barnsand Siegfried Burggraf: Crenarchaeota.
[4] J. S. Sinninghe Damsté, S. Schouten, E. C. Hopmans, A. C. T. Duin and J. A. J. Geenevasen: Crenarchaeol: the characteristic core glycerol dibiphytanyl glycerol tetraether membrane lipid of cosmopolitan pelagic crenarchaeots. J. Lipid Res. 2002, 43, pp. 1641-1651.
DOI: 10.1194/jlr.M200148-JLR200.
[5] J. S. Sinninghe Damsté, W. I. C. Rijpstra, E. C. Hopmans, F. G. Prahl, S. G. Wakeham and S. Schouten: Distribution of Membrane Lipids of Planktonic Crenarchaeota in the Arabian Sea. Appl. Env. Microbiol. 2002, 68, pp. 2997-3002.
DOI: 10.1128/AEM.68.6.2997-3002.2002.
[6] S. Schouten, E. C. Hopmans, E. Schefuß and J. S. Sinninghe Damsté: Distributional variations in marine crenarchaeotal membrane lipids: a new tool for reconstructing ancient sea water temperatures? Earth Planet Sci. Lett. 2002, 204, pp. 265-274. PDF-version.
Thursday, October 22, 2009
Acronym in microbiology: ANME for anaerobic methanotrophic archaea
The Linnaean classification system knows only two domains, plants and animals, at the highest level of its hierarchy. With the “birth” of the phylogenetic tree, archaea “arrived” as a third domain.” For details, see, for example, Archaea and the Tree of Life by Preston So.
An interesting set of microorganisms is the group (or supergroup) of anaerobic methanotrophic archaea (ANME), which are described in the glossary of the book Echoes of Life [1] as follows:
Keywords: microbes, taxonomy, phylogenetics
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; see, for example, pages 201-204 and glossary.
An interesting set of microorganisms is the group (or supergroup) of anaerobic methanotrophic archaea (ANME), which are described in the glossary of the book Echoes of Life [1] as follows:
These microorganisms participate in the anaerobic oxidation of methane and incorporate carbon from the methane. At least three phylogenetically distinct groups of ANME have been identified; these appear to be related to several genera of methanogens. ANME are often found living in close association with sulfate-reducing bacteria, sometimes in consortia where methane is oxidized and sulfate reduced.The first discovered “ANME species” was ANME 1—a name that doesn't show much resemblance to the typical binomial nomenclature used within the Linnaean system of taxonomy. With phylogenetics, the concept of a microbe species is loosing sharpness. ANME 1 was discovered and named by microbiologist Ed DeLong and his colleagues at the Monterey Bay Aquarium Research Institute in California, where they studied ribosomal RNA sequences of methanogens found in marine environments and sediments.
Keywords: microbes, taxonomy, phylogenetics
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; see, for example, pages 201-204 and glossary.
Sunday, October 18, 2009
Acronym in analytical chemistry: GC-irm-MS for gas chromatography—isotope ratio monitoring—mass spectrometry
The term gas chromatography—isotope ratio monitoring—mass spectrometry (GC-irm-MS) was coined in the 1980s in Indiana by John Hayes, Kate Freeman and others for a novel technique they applied in the determination of isotopic compositions of compounds in mixtures of environmental samples [1]. The glossary in Echoes of Life gives the following definition of the GC-irm-MS technique [1]:
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; see, for example, page 157 and glossary.
Further references to GC-irm-MS applications and results
[2] The graphical representation on page 158 in [1] gives a nice overview of differences of 13C isotopic composition found in various groups of organisms, the environment and geologic deposits.
[3] Group of 9 authors: Terrestrial vegetation change inferred from n-alkane σ13C analysis in the marine environment. Geochimica et Cosmochimica Acta 1995, 59, pp. 2853-2857.
DOI: 10.1016/0016-7047(95)00160-2.
[4] Bart E. van Dongen, Stefan Schouten and Jaap S. Sinninghe Damsté: Gas chromatography/combustion/isotope-ratio-monitoring mass spectrometeric analysis of methylboronic derivatives of monosaccharides: a new method for determining 13C abundances of carbohydrates. Rapid Communications in Mass Spectrometry 2001, 15 (7), pp. 496-500.
DOI: 10.1002/rcm.259.
A gas chromatograph combined with a combustion interface that burns the separated compounds to CO2, and with a special mass spectrometer that can then measure the relative abundance of isotopes. Used to determine the isotopic composition of individual molecular species.The acronym is sometimes also written in its all-uppercase form GC-IRM-MS. Synonymously, the acronyms irm-GC/MS and GC-IRMS are in use.
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; see, for example, page 157 and glossary.
Further references to GC-irm-MS applications and results
[2] The graphical representation on page 158 in [1] gives a nice overview of differences of 13C isotopic composition found in various groups of organisms, the environment and geologic deposits.
[3] Group of 9 authors: Terrestrial vegetation change inferred from n-alkane σ13C analysis in the marine environment. Geochimica et Cosmochimica Acta 1995, 59, pp. 2853-2857.
DOI: 10.1016/0016-7047(95)00160-2.
[4] Bart E. van Dongen, Stefan Schouten and Jaap S. Sinninghe Damsté: Gas chromatography/combustion/isotope-ratio-monitoring mass spectrometeric analysis of methylboronic derivatives of monosaccharides: a new method for determining 13C abundances of carbohydrates. Rapid Communications in Mass Spectrometry 2001, 15 (7), pp. 496-500.
DOI: 10.1002/rcm.259.
Saturday, October 17, 2009
Emily, a nickname for Emiliania huxleyi
Emily is a short term and nickname that some microbiologists like to use to refer to the coccolithophore Emiliania huxleyi, a species from a group of single-celled marine planktonic algae.
Geologists and paleoceanographers have developed much interest in Emily and related species, since Emiliania huxleyi algae produce long-chain alkenones, of which—when found in marine sediments —the ratio of molecules with different degree of unsaturation (different number of double bonds) can be used to extrapolate to past sea surface temperatures [1].
Keywords: biochemistry, paleoceanography, coccolithophores, molecular structure of unsaturated alkanones, ketones
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; see, for example, page 111 and 114.
Geologists and paleoceanographers have developed much interest in Emily and related species, since Emiliania huxleyi algae produce long-chain alkenones, of which—when found in marine sediments —the ratio of molecules with different degree of unsaturation (different number of double bonds) can be used to extrapolate to past sea surface temperatures [1].
Keywords: biochemistry, paleoceanography, coccolithophores, molecular structure of unsaturated alkanones, ketones
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; see, for example, page 111 and 114.
The term biomarker and its synonyms
The word biomarker was coined in the late 1970s by Wolfgang Seifert at Chevron, when chemical fingerprinting of sediments, crude oil and other samples with organic matter advanced rapidly due to new techniques in analytical chemistry [1]:
From a kinetics viewpoint, biomarkers are organic reaction products (or the molecules thereof)—often decomposition products of much larger biomolecules or supramolecular structures.
Keywords: organic geochemistry, biochemistry, paleontology, history
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; pp. 83-84 and glossary.
[…] Seifert was the one who coined the term “biomarker” from their original “biological marker,” perhaps because it sounded catchier, especially in an industry [oil industry] that was more interested in rocks than biology. The term caught on, even as the work with petroluem biomarkers moved farther and farther from the original concept of a biological marker—from understanding the history of life or the biological origin of hydrocarbons in petroleum, to chronicling the history of rocks and the fate of the hydrocarbons themselves.In the glossary of the book Echoes of Life [1], the term Biomarker is defined as follows:
An organic compound in natural waters, sediments, soils, fossils, crude oils, or coal that can be unambiguously linked to specific precursor molecules made by living organisms.Biological marker, molecular fossil, fossil molecule, and geochemical fossil are given as synonymous expressions for the word biomarker.
From a kinetics viewpoint, biomarkers are organic reaction products (or the molecules thereof)—often decomposition products of much larger biomolecules or supramolecular structures.
Keywords: organic geochemistry, biochemistry, paleontology, history
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; pp. 83-84 and glossary.
Friday, October 16, 2009
Dinosterol, a sterol from dinoflagellates

Dinosterol is one of many sterols that has been isolated from dinoflagellates, a genus of single-celled algae. Dinosterol was identified and named around 1978 when a Dutch group—the research team of organic chemist Jan de Leeuw at Delft University in the Netherlands— studied sterols in Black Sea sediments and another research team (Rhode Island group) analyzed natural products from marine algae that cause “red tides.”
The molecular structure of dinosterol is a biomarker in the paleontological studies of marine sediments. The book Echoes of Life [1] traces the history of the identification of such biomarker molecules. For the chemical compound dinosterol, it gives the following story:
One of its [the Dutch group's] first big successes came […] from a 5000-year-old layer of surface sediments in the Black Sea. Here the group found massive amounts of a sterol with a “peculiar” structure […]. The Black Sea sediments […] were also loaded with the distinctive fossils of dinoflagellates, a large, diverse genus of single-celled algae, and the Dutch group suspected this was the source of the sterol. In 1978, around the same time they determined its structure, a group of Rhode Island natural products chemists who were studying the toxin-producing algae [Gonyaulax tamarensis] responsible for the deadly “red tides” that periodically occur in coastal waters began isolating sterols from dinoflagellates and confirmed de Leeuw's suspicions: the most prevalent of the sterols, which they christened dinosterol, was none other than the Black Sea sterol, a hitherto unknown structure, with a methyl group attached to the A-ring, and and extra branch attached to its side chain.Synonyms for dinosterol (C30H52O), according to the Merck Index, are 4,23-dimethylergost-22-en-3-ol and—no surprise—Black Sea sterol.
Keywords: organic chemistry, sterols, algae, paleontology, history
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; page 108.
Thursday, October 15, 2009
Hopane, a pentacyclic triterpane named—indirectly—after English botanist John Hope
 Hopane and its derivatives (hopanoids) are chemical compounds that occur in certain tropical plants and also in bacterial cell membranes. Further, they have been found in rock and sediment layers that formed in inland lakes and seas, recently—on a paleontological time scale—or during the Eocene. For example, hopanes were identified in samples from Messel and Green River shales.
Hopane and its derivatives (hopanoids) are chemical compounds that occur in certain tropical plants and also in bacterial cell membranes. Further, they have been found in rock and sediment layers that formed in inland lakes and seas, recently—on a paleontological time scale—or during the Eocene. For example, hopanes were identified in samples from Messel and Green River shales.Hopane got its name from a Burmese tree, the Hopea tree, which was named after the British botanist John Hope [1]:
[…] a 30-carbon five-ring affair that people had taken to calling the “hopane” skeleton—not because it inspired particularly positive expectations, but because the genus of Burmese trees where it was first discovered was named after an eighteenth-century English botanist named John Hope. Probably the only reason anyone knew about hopane to begin with was that the British Museum has used resin from Hopea trees as a varnish. What was disconcerting was that compounds with this hopane ring structure were present in every rock or sediment sample one analyzed, no matter when or where it was formed. And yet, as far as anyone knew, such compounds were made only by a few exotic species of tropical trees, ferns, and mosses.The question that remains is Does this Burmese tree species or genus not have a Burmese (Myanmar) name? And if yes, shouldn't the name hopane change into a more Burmese-flavored name? A nightmare for chemical nomenclature!
Anyway, this wouldn't change the molecular structure of hopane (C30H52), a hydrocarbon molecule, which is shown in the sketch above with its carbon numbering and ring lettering.
Keywords: organic chemistry, botany, tree resin, history
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; page 62.
Monday, October 12, 2009
Misleading chemical names during the early days of geochemical studies of oils and shales
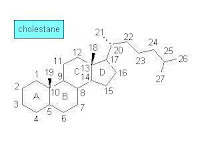 During the 1960s—the early days of GC-MS analysis in chemistry and related disciplines—a large number of natural products, obtained from both biological and geological samples, were systematically analyzed. Among the studied samples were those that included polycyclic organic compounds such as optically active terpenoids. The analytical results were leading to interesting conclusions about fossil materials and their origin while the molecular structures of compounds from living organisms and sedimentary organic matter were compared. Knowledge of the molecular structure of steroids and terpenoids became detailed enough at that time to support such comparative analysis. At first, however, some confusion in terpenoid structure and nomenclature occurred [1]:
During the 1960s—the early days of GC-MS analysis in chemistry and related disciplines—a large number of natural products, obtained from both biological and geological samples, were systematically analyzed. Among the studied samples were those that included polycyclic organic compounds such as optically active terpenoids. The analytical results were leading to interesting conclusions about fossil materials and their origin while the molecular structures of compounds from living organisms and sedimentary organic matter were compared. Knowledge of the molecular structure of steroids and terpenoids became detailed enough at that time to support such comparative analysis. At first, however, some confusion in terpenoid structure and nomenclature occurred [1]:Many of the molecular structures that they [geochemists] puzzled out did indeed resemble the optically active cyclic terpenoids that the natural products chemists were collecting from organisms—except that most of these compounds in the oils and shales had been stripped of their double bonds and oxygen-containing functional groups, reduced, as it were, to their bare carbon skeletons. Excited to find what appeared to be molecular skeletons of known biological steroids and triterpenoids, the geochemists named the hydrocarbons accordingly, and sometimes misleading—precisely because they were missing some of the key information that distinguished the biological compounds. The sterane they called “ergostane” seemed to bear the carbon skeleton of the sterol ergosterol, which is made by fungi and was quite familiar to chemists. But ergosterol differs from brassicasterol only by a double bond in the ring structure, which, of course, was missing from the steranes in the rocks, and the relatively large quantities of ergostane that the geochemists found in in rocks and oils probably had little to do with fungi and everything to do with a certain very productive group of unicellular algae. Likewise, sitosterol, which is plentiful in land plants, and stigmasterol, which is abundant in land plants and algae, are distinguished by an extra double bond in stigmasterol, so one really can't tell whether the 29-carbon sterane that geochemists first called sitostane and later stigmastane derives from land plants or algae or both. Eventually, when the geochemists gave up on trying to distinguish the orientation of the side groups in the fossil molecules, they took to calling them simply 24-methylcholestane and 24-ethylcholestane.The above sketch of the molecular structure of cholestane (C27H48) shows the carbon numbering and ring lettering used in steranes.
Keywords: paleontology, petroleum chemistry, hydrocarbons, steroids, terpenoids, steranes, alkyl-substituted cholestanes, molecular similarity, molecular difference, molecular side chains, functional groups
Reference
[1] Susan M. Gaines, Geoffrey Eglington, and Jürgen Rullkötter: Echoes of Life • What Fossil Molecules Reveal about Earth History. Oxford University Press, New York, 2009; pp. 56-57.
Behind the trademark name: Novec HFE-7100 is a mixture of the two isomers methyl nonafluorobutyl ether and methyl nonafluoroisobutyl ether
Novec™ HFE-7100 is a binary mixture of methyl perfluorobutyl ether constitutional isomers with molecular formula C5H3F9O. The components are:
Related liquids, which have been formulated for applications similar to those of Novec™ HFE-7100, are Novec™ HFE-7200 (ethoxy-nonafluorobutane), Novec™ HFE-71DE (Novec™ HFE-7100 with (E)-1,2-dichloroethene), Novec™ HFE-71DA (Novec™ HFE-7100 with (E)-1,2-dichloroethene and ethanol) and Novec™ HFE-71IPA (Novec™ HFE-7100 with 2-propanol).
- 1-methoxy-nonafluorobutane (also: methyl nonafluorobutyl ether), CF3CF2CF2CF2OCH3, CAS registry number: 163702-07-6, for physicochemical property data see ThermoML references;
- 1-methoxy-nonafluoroisobutane (also: methyl nonafluoroisobutyl ether), (CF3)2CFCF2OCH3, CAS registry number: 163702-08-7.
Related liquids, which have been formulated for applications similar to those of Novec™ HFE-7100, are Novec™ HFE-7200 (ethoxy-nonafluorobutane), Novec™ HFE-71DE (Novec™ HFE-7100 with (E)-1,2-dichloroethene), Novec™ HFE-71DA (Novec™ HFE-7100 with (E)-1,2-dichloroethene and ethanol) and Novec™ HFE-71IPA (Novec™ HFE-7100 with 2-propanol).
Friday, October 9, 2009
Acronym in chemistry: EIL for energetic ionic liquid
By combining the definitions for an ionic liquid and an energetic chemical substance, an energetic ionic liquid (EIL) is an energetic salt with a melting point below 100 °C.
Energetic compounds have a high energy content and are typically applied as fuels or explosives. Naturally, they are hazardous. Methods that allow derivatization of molecular energetic compounds into ionic ones have been suggested to provide safer routes to better manageable energetic materials.
Energetic ionic compounds such as ammonium, hydrazonium, and guanidinium salts are known for quite some time. Current design of EILs considers cations and anions that both have a high nitrogen content, typically two to four nitrogen atoms as heteroatoms in a five-membered aromatic ring with alkyl side chains as well as azido, nitro or cyano groups as substituents. The anions may also by acyclic ions such as nitrate, halides or perchlorate.
Keywords: advanced materials, molecular design, ionic liquid design, physical and chemical properties, hazardous materials, material safety
Selected literature
[1] Marcin Smigla, Andreas Metlen, and Robin D. Rogers: The Second Evolution of Ionic Liquids: From Solvents and Separations to Advanced Materials—Energetic Examples from the Ionic Liquid Cookbook. Acc. Chem. Res. 2007, 40, pp. 1182-1192.
DOI: 10.1021/ar7001304.
[2] Arindrajit Chowdhury, Stefan T. Thynell and Ping Lin: Confined rapid thermolysis/FTIR/ToF studies of tetrazolium-based energetic ionic liquids. Thermochim. Acta 2009, 485, pp. 1-3.
DOI: 10.1016/j.tca.2008.11.018.
[3] Cesar Cadena: Molecular Modeling of the Thermophysical and Transport Properties of Ionic Liquids. Dissertation, University of Notre Dame, Indiana, Sept. 2006. PDF-version.
Open access to thermodynamic data (ThermoML-encoded) for ionic liquids including EILs:
http://www.axeleratio.com/thmlbib/main/chemindexes.htm
Energetic compounds have a high energy content and are typically applied as fuels or explosives. Naturally, they are hazardous. Methods that allow derivatization of molecular energetic compounds into ionic ones have been suggested to provide safer routes to better manageable energetic materials.
Energetic ionic compounds such as ammonium, hydrazonium, and guanidinium salts are known for quite some time. Current design of EILs considers cations and anions that both have a high nitrogen content, typically two to four nitrogen atoms as heteroatoms in a five-membered aromatic ring with alkyl side chains as well as azido, nitro or cyano groups as substituents. The anions may also by acyclic ions such as nitrate, halides or perchlorate.
Keywords: advanced materials, molecular design, ionic liquid design, physical and chemical properties, hazardous materials, material safety
Selected literature
[1] Marcin Smigla, Andreas Metlen, and Robin D. Rogers: The Second Evolution of Ionic Liquids: From Solvents and Separations to Advanced Materials—Energetic Examples from the Ionic Liquid Cookbook. Acc. Chem. Res. 2007, 40, pp. 1182-1192.
DOI: 10.1021/ar7001304.
[2] Arindrajit Chowdhury, Stefan T. Thynell and Ping Lin: Confined rapid thermolysis/FTIR/ToF studies of tetrazolium-based energetic ionic liquids. Thermochim. Acta 2009, 485, pp. 1-3.
DOI: 10.1016/j.tca.2008.11.018.
[3] Cesar Cadena: Molecular Modeling of the Thermophysical and Transport Properties of Ionic Liquids. Dissertation, University of Notre Dame, Indiana, Sept. 2006. PDF-version.
Open access to thermodynamic data (ThermoML-encoded) for ionic liquids including EILs:
http://www.axeleratio.com/thmlbib/main/chemindexes.htm
Tuesday, October 6, 2009
Acronym in physics and chemistry: SHE for super heavy elements
SHE is in use as an acronym for the term super heavy elements (also written in two-word form: superheavy elements).
Uranium with atomic number 92 is a typical heavy element. To belong to the SHE set, an element needs to have a higher atomic number. Isotopes with atomic number 93 and higher are radioactive and instable—some with an extremely short life span.
SHE have super heavy atoms and, consequently, super heavy nuclei. The super heavy nucleus 298114, containing 114 protons and 184 neutrons, is often referred to as the doubly magic spherical nucleus. For nuclei in the 298114 neighborhood, enhanced nuclear stability has been predicted. The element with atomic number number 112, for which the name copernicium has been proposed, falls into this neighborhood. The term super heavy elements or its acronym SHE is most often used in the context of chemical elements that feature isotopes within this neighborhood, in which they establish islands of stability.
Uranium with atomic number 92 is a typical heavy element. To belong to the SHE set, an element needs to have a higher atomic number. Isotopes with atomic number 93 and higher are radioactive and instable—some with an extremely short life span.
SHE have super heavy atoms and, consequently, super heavy nuclei. The super heavy nucleus 298114, containing 114 protons and 184 neutrons, is often referred to as the doubly magic spherical nucleus. For nuclei in the 298114 neighborhood, enhanced nuclear stability has been predicted. The element with atomic number number 112, for which the name copernicium has been proposed, falls into this neighborhood. The term super heavy elements or its acronym SHE is most often used in the context of chemical elements that feature isotopes within this neighborhood, in which they establish islands of stability.
Monday, October 5, 2009
The name ‘copernicium’ has been suggested for the chemical element with atomic number 112
In a recent interview with Nachrichten aus der Chemie [1], physicist Sigurd Hofmann of the Center for Heavy Ion Research (GSI Helmholtz-Zentrum für Schwerionenforschung) in Darmstadt, Germany, whose international group (a total of 21 scientists) discovered and produced (a few atoms of) the element 112 in 1996, explains, why they proposed the name copernicium. He says that many names of theoretical as well as experimental physicists, who did outstanding work in atomic and nuclear physics, have already been taken and are present in the Periodic Table of Elements: for example, Nils Bohr with bohrium (element 107 with symbol Bh), Lise Meitner with meitnerium (element 109 with symbol Mt), or Wilhelm Conrad Röntgen with roentgenium (element 111 with symbol Rg). Hofman continues that this time his group wanted to go further back in history. They came across Nicolaus Copernicus, who, at the end of the middle ages and the beginning of modern times, significantly influenced the way in which we see the world (universe) today.
Symbols Cn and Cp have been proposed for element 112, assuming copernicium is going to become its name. A decision by IUPAC, which would finalize the acceptance of copernicium as the systematic element name, can be expected in 2010. Until then, the temporary name ununbium with the three-letter symbol Uub continues to be the name for this transition metal, for which isotopes with half-lifes (half-lives) in the seconds and milliseconds range have been synthesized. If synthesized in substance amount, ununbium is predicted to be liquid at room temperature, exhibiting similarity with mercury (element 80 with symbol Hg). Mercury is also known as quicksilver and shares—in the element coordination of the Periodic Table— a diagonal relationship with silver. Since the chemical element gold (Au) and ununbium also show such a diagonal relation, quickgold makes an analogical name alternative for element 112—just for the unlikely case that copernicium does not come through.
References
[1] Element 112: Ich trage einen großen Namen. Nachrichten aus der Chemie September 2009, Volume 57, page 851.
[2] ‘Copernicium’ Proposed As Name For Newly Discovered Element 122 (Science Daily) .
[3] Welcome ‘Copernicium,’ our Newest Element (Universe Today).
[4] Periodic Table of Elements including element 112 (Table annotations in German, symbols of chemical elements international).
[5] R. Eichler, W. Brüchle, R. Buda, S. Bürger, R. Dressler, Ch. E. Düllmann, J. Dvorak, K. Eberhardt, B. Eichler, C. M. Folden III, H. W. Gäggeler, K. E. Gregorich, F. Haenssler, D. C. Hoffman, H. Hummerich, E. Jäger, J. V. Kratz, B. Kuczewski, D. Liebe, D. Nayak, Nitsche, D. Piguet, Z. Qin, U. Rieth, M. Schädel, B. Schausten, E. Schimpf, A. Semchenkov, S. Soverna, R. Sudowe, N. Trautmann, P. Thörle, A. Türler, B. Wierczinski, N. Wiehl, P. A. Wilk, G. Wirth, A. B. Yakushev and A. von Zweidorf: Attempts to chemically investigate element 112. Radiochim. Acta 2006, 94, 181-191. DOI: 10.1524/ract.2006.94.4.181.
[6] R. C. Barber, H. W. Gäggeler; P. J. Karol, H. Nakahara, E. Vardaci and E. Vogt: Discovery of the Element with Atomic Number 112 (IUPAC Technical Report). Pure Appl. Chem. 2009, 81 (7), 1331-1343.
DOI: 10.1351/PAC-REP-08-03-05.
Symbols Cn and Cp have been proposed for element 112, assuming copernicium is going to become its name. A decision by IUPAC, which would finalize the acceptance of copernicium as the systematic element name, can be expected in 2010. Until then, the temporary name ununbium with the three-letter symbol Uub continues to be the name for this transition metal, for which isotopes with half-lifes (half-lives) in the seconds and milliseconds range have been synthesized. If synthesized in substance amount, ununbium is predicted to be liquid at room temperature, exhibiting similarity with mercury (element 80 with symbol Hg). Mercury is also known as quicksilver and shares—in the element coordination of the Periodic Table— a diagonal relationship with silver. Since the chemical element gold (Au) and ununbium also show such a diagonal relation, quickgold makes an analogical name alternative for element 112—just for the unlikely case that copernicium does not come through.
References
[1] Element 112: Ich trage einen großen Namen. Nachrichten aus der Chemie September 2009, Volume 57, page 851.
[2] ‘Copernicium’ Proposed As Name For Newly Discovered Element 122 (Science Daily) .
[3] Welcome ‘Copernicium,’ our Newest Element (Universe Today).
[4] Periodic Table of Elements including element 112 (Table annotations in German, symbols of chemical elements international).
[5] R. Eichler, W. Brüchle, R. Buda, S. Bürger, R. Dressler, Ch. E. Düllmann, J. Dvorak, K. Eberhardt, B. Eichler, C. M. Folden III, H. W. Gäggeler, K. E. Gregorich, F. Haenssler, D. C. Hoffman, H. Hummerich, E. Jäger, J. V. Kratz, B. Kuczewski, D. Liebe, D. Nayak, Nitsche, D. Piguet, Z. Qin, U. Rieth, M. Schädel, B. Schausten, E. Schimpf, A. Semchenkov, S. Soverna, R. Sudowe, N. Trautmann, P. Thörle, A. Türler, B. Wierczinski, N. Wiehl, P. A. Wilk, G. Wirth, A. B. Yakushev and A. von Zweidorf: Attempts to chemically investigate element 112. Radiochim. Acta 2006, 94, 181-191. DOI: 10.1524/ract.2006.94.4.181.
[6] R. C. Barber, H. W. Gäggeler; P. J. Karol, H. Nakahara, E. Vardaci and E. Vogt: Discovery of the Element with Atomic Number 112 (IUPAC Technical Report). Pure Appl. Chem. 2009, 81 (7), 1331-1343.
DOI: 10.1351/PAC-REP-08-03-05.
Tuesday, September 29, 2009
Early names for Lake Tahoe: Mountain Lake, Lake Bigler, Lake Bonpland
The name of Lake Tahoe derives from the word the Washoe Indians used to name the lake: da ow a ga. Walking the Eagle Trail Loop near the Upper Eagle Falls above Emerald Bay at the southwestern part of Lake Tahoe in California, you can find an interpretative board that mentions a few other names the lake had, before it became the well-known tourist destination it is today: When Lieutenant John Charles Frémont (see A common place-name of the West: Fremont) and Charles Preuss sighted the lake from a ridge near Carson pass on Valentine's Day, 1844, while attempting a winter crossing of the Sierra Nevada (all still Mexican territory then), they originally named it Lake Bonpland, although early maps also reflect the name Mountain Lake. At high elevation, located between Californian mountain peaks of the Sierra Nevada and the Carson Range of Nevada, the latter was an appropriate name for this alpine lake. However, in 1852 the California legislature renamed it to Lake Bigler, after John Bigler, the 3rd governor of California. After 1860, the Washoe-language-based name, Tahoe, became popular and Lake Tahoe has since resisted any further name changes.
Keywords: history, geography, place-name origins, place-name synonyms
Keywords: history, geography, place-name origins, place-name synonyms
Friday, September 25, 2009
Petroleomics, petroleome, and their counterparts in protein chemistry
The term petroleomics refers to the chemical study of petroleum including its physicochemical characterization and the identification of its molecular constituents. The noun petroleomics reminds us of the word proteomics in protein chemistry standing for the study of proteins including their molecular and supramolecular structures and functions.
The term petroleome refers to the entire amount of a petroleum sample. The goal is to know the complete chemical composition of petroleomes. Again, an analogous word for a similar concept in protein chemistry comes to mind: proteome, composed from protein and genome.
The word petroleomics was coined by Alan G. Marshall at the National High Magnetic Field Laboratory at Florida State University, where he and his group is working on analytical methods for the compositional characterization and molecular description of petroleum fractions such as asphaltenes [1].
Keywords: petrochemistry, word analogy
References and literature
[1] Celia Henry Arnaud: Digging Into Asphaltenes • Mass spectrometry uncovers chemical details of petroleum's most recalcitrant fraction. Chemical & Engineering News September 21, 2009, Volume 87, Number 38, pp. 12-17.
[2] Oliver C. Mullins, Eric Y. Sheu, Ahmed Hammani and Alan G. Marshall: Asphaltenes, Heavy Oils, and Petroleomics. Springer, New York, 2007. DOI: 10.1007/0-387-68903-6 (PDF versions of all chapters).
[3] Oliver C. Mullins: Petroleomics and Structure-Function Relations of Crude Oils and Asphaltenes (Chapter 1 in [2]).
DOI: 10.1007/0-387-68903-6_1.
The term petroleome refers to the entire amount of a petroleum sample. The goal is to know the complete chemical composition of petroleomes. Again, an analogous word for a similar concept in protein chemistry comes to mind: proteome, composed from protein and genome.
The word petroleomics was coined by Alan G. Marshall at the National High Magnetic Field Laboratory at Florida State University, where he and his group is working on analytical methods for the compositional characterization and molecular description of petroleum fractions such as asphaltenes [1].
Keywords: petrochemistry, word analogy
References and literature
[1] Celia Henry Arnaud: Digging Into Asphaltenes • Mass spectrometry uncovers chemical details of petroleum's most recalcitrant fraction. Chemical & Engineering News September 21, 2009, Volume 87, Number 38, pp. 12-17.
[2] Oliver C. Mullins, Eric Y. Sheu, Ahmed Hammani and Alan G. Marshall: Asphaltenes, Heavy Oils, and Petroleomics. Springer, New York, 2007. DOI: 10.1007/0-387-68903-6 (PDF versions of all chapters).
[3] Oliver C. Mullins: Petroleomics and Structure-Function Relations of Crude Oils and Asphaltenes (Chapter 1 in [2]).
DOI: 10.1007/0-387-68903-6_1.
A sobriquet for asphaltene: “cholesterol of petroleum”
While cholesterol promotes clogging of arteries in biological systems, asphaltene can cause clogging of pipes in refining facilities and of pores and channels in geochemical systems such as underground rocks of oil-field reservoirs. Hence, the sobriquet “cholesterol of petroleum” for asphaltenes [1]. These are complex mixtures consisting of aromatic and heterocyclic compounds. Asphaltenes occur in crude oil along with saturated, unsaturated, and aromatic hydrocarbons and hetero-functionalized derivatives thereof.
The “19th-century definition” of asphaltenes, obtained as petroleum fractions, focuses on the solubility of asphaltene constituents in other organic solvents [1]: The asphaltene fraction comprises components that are soluble in toluene but insoluble in n-heptane or n-pentane.
To model and predict asphaltene properties such as as viscosity and aggregation [2] behavior and to optimize petroleum processing operations, a detailed molecular understanding of asphaltene is required. The “cholesterol-like” activity of asphaltene depends on intra- and extramolecular interactions of its molecular constituents.
Keywords: petrochemistry, thermodynamics of petroleum, nickname of petroleum fraction, analogy
References
[1] Celia Henry Arnaud: Digging Into Asphaltenes • Mass spectrometry uncovers chemical details of petroleum's most recalcitrant fraction. Chemical & Engineering News September 21, 2009, Volume 87, Number 38, pp. 12-17.
[2] Narve Aske, Harald Kallevik, Einar Eng Johnson and Johan Sjöblom: Asphaltene Aggregates from Crude Oil and Model Systems Studied by High-Pressure NIR Spectroscopy. Energy & Fuels 2002, 16, pp. 1287-1295. DOI: 10.1021/ef020065i.
The “19th-century definition” of asphaltenes, obtained as petroleum fractions, focuses on the solubility of asphaltene constituents in other organic solvents [1]: The asphaltene fraction comprises components that are soluble in toluene but insoluble in n-heptane or n-pentane.
To model and predict asphaltene properties such as as viscosity and aggregation [2] behavior and to optimize petroleum processing operations, a detailed molecular understanding of asphaltene is required. The “cholesterol-like” activity of asphaltene depends on intra- and extramolecular interactions of its molecular constituents.
Keywords: petrochemistry, thermodynamics of petroleum, nickname of petroleum fraction, analogy
References
[1] Celia Henry Arnaud: Digging Into Asphaltenes • Mass spectrometry uncovers chemical details of petroleum's most recalcitrant fraction. Chemical & Engineering News September 21, 2009, Volume 87, Number 38, pp. 12-17.
[2] Narve Aske, Harald Kallevik, Einar Eng Johnson and Johan Sjöblom: Asphaltene Aggregates from Crude Oil and Model Systems Studied by High-Pressure NIR Spectroscopy. Energy & Fuels 2002, 16, pp. 1287-1295. DOI: 10.1021/ef020065i.
Thursday, September 24, 2009
A common place-name of the West: Fremont
Browsing a map of California, it is not hard to find a place-name that has the word Fremont in it. This name refers to John Charles Frémont, who literally put many parts of the western United States on the map. When he was born in Savannah, Georgia, on January 21, 1813, his name was spelled John Charles Fremon [1]. His father was the French teacher Charles Fremon. John Charles grew up in Charleston, South Carolina, where he adopted the french spelling of his name: Frémont. He became a lieutenant in the army corps of engineers in Washington and was sent west for surveying expeditions during the time when James Knox Polk was the president of the United States. Walter Borneman summarizes Frémont's westward drift as follows [1]:
Reference
[1] Walter R. Borneman: Polk • The Man Who Transformed the Presidency and America. Random House Trade Paperback Edition, New York, 2009; pages 182-184.
John Charles Frémont was, and remains, something of an enigma. To his defenders, Frémont will always be “the Pathfinder of the West,” the quintessential explorer marching westward, ever westward. To his detractors, Frémont was an opportunistic bungler, a man who—had it not been for the political connections and journalistic talents of his wife and the dedicated services of his mountain man guides, including Kit Carson—might have simply marched off history's map.The acute accent over the letter e in his name is now often dropped in American-English writings. If still alive, Frémont would not be pleased!
As usual with someone who elicits such strong and divergent passions, the truth lies somewhere in between. Certainly, there is no denying that the West is covered with place-names—Frémont peaks, lakes, rivers, towns, and counties— that mark his paths.
Reference
[1] Walter R. Borneman: Polk • The Man Who Transformed the Presidency and America. Random House Trade Paperback Edition, New York, 2009; pages 182-184.
Monday, September 21, 2009
A new English verb from the German language: to hocker
Hockern is a new recreational activity on the rise in Germany and elsewhere. The German verb hockern is derived from the masculine noun Hocker, meaning milking stool. Hockern can be translated into English as to busy oneself with a milking stool or to do tricks with a milking stool. To simplify translations, I would suggest to use hocker—as a regular verb—in English. It rhymes with soccer and is inflected in the same way as the verb tinker: He hockers. She hockered. They are hockering. Since the vowel e is unstressed, there is no doubling of the consonant r.
A person that hockers, may be called a hockerer or hocker gymnast.
If you are not yet convinced that hockern is a new sport, which needs to be verb-ized into English, you may just want to sit on your Hocker and watch while others are hockering along.
Keywords: creativity, gymnastics, body dynamics, fun; linguistics, English words, German words
More on Hockern
Hockern / Sport with a German Milking Stool, Feb. 06, 2008.
Hockern in Frankfurt... (video)
Kieler Kneipensport "Hockern" (Spiegel Online)
Freizeit: Locker vom Hocker (Märkische Allgemeine, September 12, 2009)
A person that hockers, may be called a hockerer or hocker gymnast.
If you are not yet convinced that hockern is a new sport, which needs to be verb-ized into English, you may just want to sit on your Hocker and watch while others are hockering along.
Keywords: creativity, gymnastics, body dynamics, fun; linguistics, English words, German words
More on Hockern
Hockern / Sport with a German Milking Stool, Feb. 06, 2008.
Hockern in Frankfurt... (video)
Kieler Kneipensport "Hockern" (Spiegel Online)
Freizeit: Locker vom Hocker (Märkische Allgemeine, September 12, 2009)
Saturday, September 19, 2009
Group-theoretical confusion, rhyming words and non-rhyming names
The words rage and age definitely rhyme. The pairs rhyme–time and group–loop are spelled differently, but still do rhyme. We expect rhyming words in songs and rhymes. Sometimes, however, the fact that words do not rhyme at line's end is intended. The following song, that captures a panic during an epoch of exciting discoveries in group theory [1], is a case in point:
The panic of the 1960s and 1970s is over, unless you fear the Monster. Now finite simple groups are classified (completely?) and group theorists have their Atlas of Finite Groups, also known as Atlas of Symmetry (a “Rosetta Stone of science”), where groups are mapped out by beginning with A5 and finishing with M, the Monster that might as well been called the Friendly Giant or the Fischer-Griess group (see page 336 in [1]).
The names of mathematicians, mentioned in the above song, refer to Zvonimir Janko, John Horton Conway, Bernd Fischer, Dieter Held, Jack McLaughlin, Michio Suzuki, Graham Donald Higman, and Charles Sims.
Keywords: rhyme, mathematicians, symmetry, history of group theory, classification of finite simple groups
Reference
[1] Marcus du Sautoy: Symmetry. A Journey into the Patterns of Nature. First Harper Perennial Edition, Harper Collins Publishers, New York, 2009; page 306.
The floodgates were opened! New groups were the rage!
(And twelve or more sprouted, to greet the new age.)
By Janko and Conway and Fischer and Held
Mc Laughlin, Suzuki, and Higman, and Sims.
No doubt you noted the last lines don't rhyme.
Well, that is, quite simply, a sign of the time.
There's chaos, not order, among simple groups;
And maybe we'd better go back to the loops.
The panic of the 1960s and 1970s is over, unless you fear the Monster. Now finite simple groups are classified (completely?) and group theorists have their Atlas of Finite Groups, also known as Atlas of Symmetry (a “Rosetta Stone of science”), where groups are mapped out by beginning with A5 and finishing with M, the Monster that might as well been called the Friendly Giant or the Fischer-Griess group (see page 336 in [1]).
The names of mathematicians, mentioned in the above song, refer to Zvonimir Janko, John Horton Conway, Bernd Fischer, Dieter Held, Jack McLaughlin, Michio Suzuki, Graham Donald Higman, and Charles Sims.
Keywords: rhyme, mathematicians, symmetry, history of group theory, classification of finite simple groups
Reference
[1] Marcus du Sautoy: Symmetry. A Journey into the Patterns of Nature. First Harper Perennial Edition, Harper Collins Publishers, New York, 2009; page 306.
Wednesday, September 16, 2009
Abbreviation: Trhd. for trailhead
 The composed noun trailhead is sometimes abbreviated as trhd. My example shows a sign at the end of the Jobs Peak Ranch Trail in the eastern part of Douglas County, Nevada. If you arrived here by coming from the Jobs Peak Ranch Trailhead, it will take you another 1.4 miles to return to where you started. Instead, looking in the other direction, a sign informs you that you may continue on to reach the Fay-Luther Trhd. after 2.0 miles. In fact, the units in miles have been omitted on both signs; probably another form of abbreviating.
The composed noun trailhead is sometimes abbreviated as trhd. My example shows a sign at the end of the Jobs Peak Ranch Trail in the eastern part of Douglas County, Nevada. If you arrived here by coming from the Jobs Peak Ranch Trailhead, it will take you another 1.4 miles to return to where you started. Instead, looking in the other direction, a sign informs you that you may continue on to reach the Fay-Luther Trhd. after 2.0 miles. In fact, the units in miles have been omitted on both signs; probably another form of abbreviating.
Saturday, September 12, 2009
Great dodecahedron versus Platonic dodecahedron
The dodecahedron is a Platonic solid along long with the tetrahedron, cube, octahedron and icosahedron. The faces of these five highly symmetric, three-dimensional objects are regular polygons (equilateral triangle, square and regular pentagon). A “spherical composition” of a specific number of same-type polygons (12 pentagons in the dodecahedron case) forms a Platonic solid. Each pair of adjacent polygons share an edge. The building polygons are not allowed to intersect, according to a condition imposed by Theaetetus, a colleague of the philosopher Plato, after which the Platonic solids are named. But what if they are allowed to intersect? Marcus du Sautoy “answers” this question with the following historical observation [1]:
Whatever they are named, there are only four of them as Augustin-Louis Cauchy (1789-1857) proved by successfully answering the question of the French Academy of Science (founded in 1666), which had dedicated a prize in 1811 for “proving beyond doubt that the five Platonic solids plus the four new solids were all the three-dimensional shapes that you could build from identical regular polygons” [1].
Keywords: solid geometry, nomenclature of polydedra
References and further reading and visualizing
[1] Marcus du Sautoy: Symmetry. A Journey into the Patterns of Nature. First Harper Perennial Edition, Harper Collins Publishers, New York, 2009; pages 164-166.
[2] Chapter VI “Star-Polyhedra” in the book by H. S. M. Coxeter: Regular Polytopes. Dover Publications, New York, 1973.
[3] Image of a great dodecahedron and more at Wolfram
To everyone's surprise, in 1809 a new shape had been built out of these 12 pentagons [that were known to build the Platonic dodecahedron]. Theaetetus had insisted that the faces of his shapes should not cut into each other. But what if you relaxed this condition? A mathematics teacher in Paris had found a new way to piece 12 pentagons together to make a new symmetrical shape that was christened the great dodecahedron. Although it looks like a shape built from lots of irregular triangles, it consists of 12 intersecting pentagons. The shape satisfies all the conditions for a Platonic solid except for the fact that the faces cut into each other. How many other strange and beautiful shapes like this might be out there? Three others were soon discovered, and mathematicians began to wonder where the new list might end.Should these polyhedra with intersecting regular polygons be named anti-Theaetetian solids? Since they look star-shaped, they are otherwise addressed as stellated polyhedra [2].
Whatever they are named, there are only four of them as Augustin-Louis Cauchy (1789-1857) proved by successfully answering the question of the French Academy of Science (founded in 1666), which had dedicated a prize in 1811 for “proving beyond doubt that the five Platonic solids plus the four new solids were all the three-dimensional shapes that you could build from identical regular polygons” [1].
Keywords: solid geometry, nomenclature of polydedra
References and further reading and visualizing
[1] Marcus du Sautoy: Symmetry. A Journey into the Patterns of Nature. First Harper Perennial Edition, Harper Collins Publishers, New York, 2009; pages 164-166.
[2] Chapter VI “Star-Polyhedra” in the book by H. S. M. Coxeter: Regular Polytopes. Dover Publications, New York, 1973.
[3] Image of a great dodecahedron and more at Wolfram
Friday, September 11, 2009
An Argand diagram is a Wessel diagram is a Gauss diagram
An Argand diagram is a two-dimensional coordinate system for the presentation of complex numbers. The scaling on the horizontal axis gives the real part and the scaling on the vertical axis gives the imaginary part of a complex number. Although Jean-Robert Argand (1768-1822) is usually credited for geometrically picturing complex numbers this way, Marcus du Sautoy provides a detailed look into the origin of complex number presentation [1]:
Hence, the terms Wessel diagram or Gauss diagram would as well be justified.
Instead of using the word diagram for a figure, that illustrates complex numbers and their relationships graphically, the word plane is often used: Argand plane instead of Argand diagram. Other synonymous terms for Argand plane are complex plane or z-plane; giving credit to the complex numbers z = a + bi again!
Keywords: mathematics, geometry, algebra, history, graphical illustration, synonymous expressions
References
[1] Marcus du Sautoy: Symmetry. A Journey into the Patterns of Nature. First Harper Perennial Edition, Harper Collins Publishers, New York, 2009; page 153.
[2] Orlando Merino: A Short History of Complex Numbers. University of Rhode Island, 2006; PDF-file.
Gauss [Carl Friedrich, (1777-1855)] had actually used a picture of the imaginary numbers as a mathematical tool in his proof [of the Fundamental Theorem of Algebra], but he kept it hidden for many years, fearing he would be laughed at by a mathematical establishment still wedded to the language of equations and formulae. But because the image was so powerful and gave imaginary numbers a physical reality, it was only a matter of time before others hit upon the idea. Two amateur mathematicians, the Dane [/Norwegian] Caspar Wessel [(1745-1818)] and the Swiss Jean Argand, independently proposed similar pictures in pamphlets they published. Argand, who was the last of the three to have the idea, is the person whose name became attached to the picture we now call the Argand diagram. Credit is rarely just.
Hence, the terms Wessel diagram or Gauss diagram would as well be justified.
Instead of using the word diagram for a figure, that illustrates complex numbers and their relationships graphically, the word plane is often used: Argand plane instead of Argand diagram. Other synonymous terms for Argand plane are complex plane or z-plane; giving credit to the complex numbers z = a + bi again!
Keywords: mathematics, geometry, algebra, history, graphical illustration, synonymous expressions
References
[1] Marcus du Sautoy: Symmetry. A Journey into the Patterns of Nature. First Harper Perennial Edition, Harper Collins Publishers, New York, 2009; page 153.
[2] Orlando Merino: A Short History of Complex Numbers. University of Rhode Island, 2006; PDF-file.
Thursday, September 10, 2009
The word “via” as noun and preposition
The word via has a Latin origin, meaning way or road as in Via Appia (Appian Way), which is the name of a Roman road that connected Rome to Brindisi in what is now southern Italy.
In English, this meaning of connecting is still present when via is used as a noun—even on a microscale. In microelectronic engineering, for example, the short word via is used to name an interconnect hole or through-hole on an integrated circuit. A via connects two conductive layers separated by a dielectric layer. Vias are typically etched through dielectric layers and then filled with a conductive metal to make an electrical connection.
The word via is also used as a preposition with meanings by way of or by means of (as in sending via email). With via, one can emphasize an applied process (as in separating via distillation). Typically, via can be replaced by prepositions such as by, through, or with with a slight loss of nuance. Also, a detour or unexpected route is often expressed via via: She came from Cyprus via Canada to Chicago.
In English, this meaning of connecting is still present when via is used as a noun—even on a microscale. In microelectronic engineering, for example, the short word via is used to name an interconnect hole or through-hole on an integrated circuit. A via connects two conductive layers separated by a dielectric layer. Vias are typically etched through dielectric layers and then filled with a conductive metal to make an electrical connection.
The word via is also used as a preposition with meanings by way of or by means of (as in sending via email). With via, one can emphasize an applied process (as in separating via distillation). Typically, via can be replaced by prepositions such as by, through, or with with a slight loss of nuance. Also, a detour or unexpected route is often expressed via via: She came from Cyprus via Canada to Chicago.
Wednesday, September 9, 2009
Synonyms in mathematics: remainder and residue
The remainder is what is “left over” when an integer is divided by a non-zero integer. For example, the division of 11 by 7 results in 1 and leaves the remainder 4. In the terminology of number theory, this can be written as 11 ≡ 4 mod 7 (in words: 11 and 4 are congruent modulo 7 or, synonymously, 4 is a residue of 11 modulo 7) .
In general notation: a ≡ b mod m, expressing that the difference a-b is an integral multiple of m. The symbol b represents the remainder or residue.
Note: The German word for remainder or residue is the masculine noun Rest. Infrequently, the word rest can be found in English-language math texts, used as a synonym for remainder or residue.
Keywords: arithmetics, mathematics, number theory, division, English language, German language
In general notation: a ≡ b mod m, expressing that the difference a-b is an integral multiple of m. The symbol b represents the remainder or residue.
Note: The German word for remainder or residue is the masculine noun Rest. Infrequently, the word rest can be found in English-language math texts, used as a synonym for remainder or residue.
Keywords: arithmetics, mathematics, number theory, division, English language, German language
Sunday, September 6, 2009
Symmetry from the Greek words “syn” and “metros”
The five regular polyhedra (tetrahedron, hexahedron or cube, octahedron, dodecahedron and icosahedron) are often cited as an expression of perfect symmetry. These polyhedra are today called Platonic solids after the Greek Philosopher Plato. He thought they were so fundamental that they must be the basic building units shaping the material world. The word symmetry began shaping at around the same time, when Plato founded his science and philosophy institution in Athens in 387 BC. In his book about symmetry [1], Marcus du Sautoy explains how the word symmetry made its way into the English language, from Greek via Latin:
Keywords: geometry, mathematics, etymology, history, Greek, Latin
Reference
[1] Marcus du Sautoy: Symmetry. A Journey into the Patterns of Nature. First Harper Perennial Edition, Harper Collins Publishers, New York, 2009.
It is the Greek language that assigned a name to the common trait that bound Plato's five objects [Platonic solids] together: symmetros. In the first century AD the Roman author Pliny the Elder bemoaned Latin's lack of a word for symmetry. Symmetros combines the Greek words syn, meaning ‘same’, and metros, meaning ‘measure’. Together they describe something ‘ with equal measure’. Symmetry for the Greeks was reserved for describing an object in which some of the internal physical dimensions were the same across the shape. In symmetrical solids the edges were all the same length, the faces all had the same area, and the angles between adjacent faces were all equal. Symmetry is about measurement and geometry. It would take some time for symmetry to become recognized as a mathematical property that goes beyond simple measurement, although the Greek philosophers were beginning to explore the idea of symmetry as a powerful image beyond physical shapes.
Keywords: geometry, mathematics, etymology, history, Greek, Latin
Reference
[1] Marcus du Sautoy: Symmetry. A Journey into the Patterns of Nature. First Harper Perennial Edition, Harper Collins Publishers, New York, 2009.
Subscribe to:
Posts (Atom)


